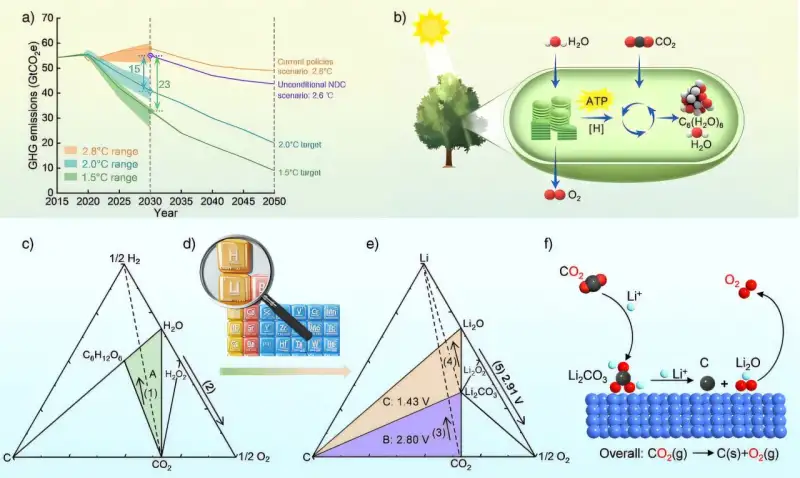
Imagine converting the very gas driving climate change into something as vital as oxygen. A team of Chinese researchers has taken a bold step toward this vision by developing a highly efficient electrochemical method to split carbon dioxide (CO₂) into pure oxygen and elemental carbon. Reported in the journal Angewandte Chemie International Edition, this breakthrough achieves an astonishing oxygen yield of 98.6%, and its implications could stretch from saving Earth’s atmosphere to sustaining life on distant planets.
At the heart of this innovation is a cleverly designed system that uses lithium as a mediator and a ruthenium-cobalt (RuCo) catalyst to perform a controlled two-step transformation. In the first step, CO₂ reacts with lithium inside an electrochemical cell, forming lithium carbonate (Li₂CO₃). In the next stage, this compound is broken down to generate lithium oxide (Li₂O) and solid carbon. Then, through oxidation, lithium oxide releases oxygen gas and regenerates lithium ions, ready to start the process all over again. The RuCo catalyst ensures that each step happens efficiently, minimizing energy loss—an essential quality for any viable environmental or space technology.
What makes this discovery so exciting isn’t just its efficiency but its adaptability. The team tested the method under various atmospheric conditions—including pure CO₂, simulated flue gas mixtures like those produced by power plants, and even a Mars-like environment containing just 1% CO₂ in argon. The system held strong across all these tests, consistently generating high-purity oxygen. This versatility makes the technology an outstanding candidate for supporting long-duration space missions, where carrying large oxygen reserves is impractical. For future Mars missions, this system could mean astronauts generate breathable air directly from the Martian atmosphere.
Here on Earth, the implications are just as powerful. If powered using renewable energy, this process could serve as a carbon-neutral or even carbon-negative solution for industries grappling with CO₂ emissions. Instead of merely capturing CO₂ for storage, we could transform it into usable oxygen while simultaneously locking carbon into solid form. This opens doors for diverse applications—oxygen production for underwater habitats, closed-loop life support systems, personal breathing equipment, emergency survival gear, and even industrial gas treatment systems.
The research, led by Ping He and Haoshen Zhou at Nanjing University with collaborators from Fudan University, highlights a path forward that bridges environmental responsibility and technological advancement. The authors believe their work could redefine how we address planetary sustainability and how we prepare for life beyond Earth. As highlighted by ScienceDaily, the method’s ability to perform in both terrestrial and extraterrestrial environments could make it a cornerstone in future space colonization efforts.
Perhaps the most inspiring aspect of this breakthrough is its elegance. Unlike high-temperature or high-pressure alternatives, this method operates electrochemically at milder conditions, relying on clever chemistry and catalysis rather than brute force. And while many CO₂ conversion techniques suffer from low selectivity or require high energy input, this approach stands out for its purity of products and cyclic efficiency.
In essence, this is more than just a new reaction. It’s a vision of a future where CO₂ isn’t just a pollutant to trap or bury, but a resource to harness—where a harmful greenhouse gas becomes a source of breathable air. As the world faces mounting pressure to decarbonize, technologies like this could tip the balance, offering a cleaner, more sustainable way to manage the atmosphere. And as humanity sets its sights on Mars and beyond, the ability to turn CO₂ into life-giving oxygen could be the difference between surviving and thriving in the cosmos.